Last Updated November 5, 2020
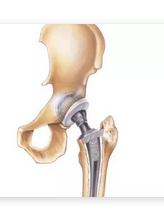
In the United States, the number of Total Hip Arthroplasty (THA) procedures performed in recent years has increased substantially – especially in a growing number of young patients. Physicians and clinicians believe hip replacement surgery is one of the most cost-effective ways to improve the quality of life in suffering patients. However, although there are generally many benefits to suffering patients from hip replacement surgery – as with any invasive procedure, there are risks and the possibility for complications.
The vast majority of patients who undergo THA find significant pain relief and improvement in mobility following the procedure. Bearing that in mind, there are factors such as patient obesity or other medical conditions such as diabetes, liver disease, heart disease, or other uncemented prostheses which may contribute negatively to the postoperative outcome. Patients are always recommended to develop an open line of communication with their physicians and surgeons to identify specific risks prior to surgery and possibly undertake lifestyle modifications such as quitting smoking or losing weight.
The bottom line for both doctors and patients is to avoid unfortunate complications which can have life-altering consequences as well as the need for follow-up surgical procedures known as “revision” surgery. Below are some of the more significant and unwelcome risks associated with hip replacement surgery:
Blood Clots
Deep Vein Thrombosis (DVT) is a widely recognized risk factor following hip replacement surgery. DVT is a blood clot in the leg that can break loose and become a Pulmonary Embolism (PE) that travels through the body and into the lungs where it can cause illness and death. DVT can occur in any vein in the body, however, they tend to manifest the most often in the lower extremities of the body.
Symptoms of DVT may not be obvious or even recognizable to the patient. In fact, DVTs can occur with no symptoms at all about half of the time. When there are visible symptoms, they usually look/feel like:
- Swelling in the leg
- Red, discolored, or white skin
- A cord in a leg vein that can be felt
- Rapid heartbeat
- Slight fever
- Warm skin
- More visible surface veins
- A dull ache, tightness, tenderness, or pain in the leg
Any patient with symptoms that concern them should call their physician immediately.
Risk factors for DVT or that may contribute to DVT include obesity, inherited traits that cause clotting, being older than 60, and having Type-A blood.
Patients can take steps to prevent DVT following surgery as well and physicians may prescribe exercise/physical therapy, compression stockings, and anti-clotting medicines to help combat the risk.
Hip Dislocation
Traumatic hip dislocation occurs when the ball of the hip joint is pushed out of the socket and is usually the result of an incident such as an auto collision or a high-impact fall. A dislocated hip can result in life-changing and long-term issues that can be debilitating if not addressed promptly.
Hip dislocation following THA is relatively infrequent among healthy people under the close guidance and care of a physician. However, there tend to be higher rates of dislocation among some sets of hip replacement patients, such as the elderly or those who have suffered from previous hip injuries.
If a patient or physician suspects that a hip joint has dislocated, they will typically conduct a physical exam and order imaging of the hip with either a CT scan or MRI. An orthopaedist usually can manipulate the hip-joint back into place while the patient is under anesthesia. However, if the damage appears to be more severe, revision surgery may be necessary.
Infection
Infection following surgery for THA is a possibility and a very serious problem. Accidental exposure to bacteria such as staphylococcus aureus can occur even in the midst of the most stringent sterilization protocols.
People who live with certain conditions, such as diabetes mellitus; obesity; peripheral vascular disease; or are otherwise immunocompromised; are at greater risk for developing infections even in a hospital setting.
Common symptoms of joint-infection following surgery include:
- Increased stiffness and pain
- Swelling
- Redness
- Wound drainage
- Fevers, nightsweats and chills
- Fatigue
Treatment options for joint-infection depend upon the extent and depth of the infection. Superficial infections in soft tissue are relatively easy to treat with oral antibiotics or even intravenous (IV) antibiotics. For deeper infections, including those within the joint itself, treatment may require surgical treatments such as debridement – or removal of the infected soft tissue and thorough cleaning of implanted devices and spacers.
Damage to Joint Structures
There exists a distinct possibility that soft muscle and joint tissue surrounding the hip will be damaged during surgery. This damage, in some cases, can result in leg weakness or decreased feeling in the leg.
Loosening of the Prosthetic
A new hip joint should fit naturally with the existing bone and surrounding tissue. However, over time, one or both ends of the replaced joint may loosen the bond to the natural bone causing pain or other problems.
When a hip replacement is implanted, it is either pressed or cemented into position to fit tightly into the bone of the thigh or pelvis. A hip replacement may start to loosen due to factors involving: weight, age, sex, and activity levels. Statistics have shown that women, people over 60, and those with a Body Mass Index (BMI) under 25 tend to have the least amount of issues with loosening.
The most common issue connected with the loosening of the hip joint is Osteolysis – a condition akin to the bone around the implant seemingly “melting away”. Because of the weakened bone around the hip joint, it begins to come loose and wobble.
Loosening issues may necessitate a repair surgery known as a Hip Revision. Revision surgery may also be recommended if an infection has developed in the tissue surrounding the joint.
Femur Fracture
Bone fractures in combination with prosthetic joint replacements are referred to as “periprosthetic fractures”. They are generally rare but are a very serious complication of hip replacement surgery. These types of fractures usually occur years after surgery and can be challenging to repair due to the age of the patient. They are usually the result of a fall or other similar trauma. Most cases of periprosthetic fractures require surgery to treat.

Metal-on-Metal Implants and Metallosis
In Metal-on-Metal (MoM) hip implants, the metal ball and metal cup slide against each other during walking or running. This friction between the ball and socket can cause the release of metal particles that will wear off of the device and embed in surrounding tissue as well as metal ions of cobalt and chromium entering the bloodstream. Over time, this leaching of metal from the joint into surrounding tissue causes “Adverse Local Tissue Reaction” (ALTR) and/or “Adverse Reaction to Metal Debris” (ARMD). The bottom line for patients implanted with MoM joints, however, is that they suffer damage and pain, device failure, loosening, and the need for revision surgery. Furthermore, patients implanted with MoM hip joints can also suffer from a condition known as “metallosis” – a potentially fatal complication arising from metallic erosion in the joint which induces pain around the joint, pseudotumors, and a noticeable rash.
Prior to 2016, MoM hip devices marketed in the United States were subject to the FDA’s less rigorous 510(k) clearance process which allowed them to be implanted in human patients without first subjecting them to the same standards of scientific review and lab testing as the FDA’s more rigorous Pre-Market Approval (PMA) process. In May 2016, following numerous recommendations, the FDA required all MoM manufacturers to immediately cease and desist from marketing MoM devices until they can be demonstrably approved under PMA conditions with valid supporting scientific evidence. It is worth noting that to-date, there are no FDA-approved MoM hip replacement devices marketed for use in the United States.
Hip Revision
Despite the overall favorable statistics concerning THA, hip replacement surgery can still fail for a number of reasons. When hip replacement failure occurs, a physician may recommend a second procedure to examine or even remove some of a patient’s prosthesis and replace it with a new device. This second procedure is known as a “revision”.
The hip revision could turn out to be a longer and more complex procedure than even the first surgery. Additionally, there are different types of hip revision surgery depending upon the extent of injury or replacement. In some cases, only parts of a prosthetic joint may be addressed, while in another revision – the entire joint may be replaced.
As discussed earlier, a physician may order a revision on the basis of a range of circumstances, including infection; fracture; loosening; dislocation, or reaction to metallosis. Revision surgery carries similar risks as primary hip replacement surgery. However, the success rate for revision surgery is generally lower due to issues with the structural strength of the bone.
Patients undergoing revision surgery can take certain steps to minimize risks in advance, such as:
- Avoiding overuse of the joint
- Avoiding sports which involve jumping
- Maintaining a healthy weight
Following revision surgery, patients should keep in close contact with their physician and surgeon if additional pain develops suddenly or it appears that an infection has developed.
Sources Cited (43)
1) “Complications of Total Hip Arthroplasty: Standardized List, Definitions, and Stratification Developed by The Hip Society” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4709292/#:~:text=Based%20on%20this%20work%20from,instability%2C%20periprosthetic%20fracture%2C%20abductor%20muscle
2) “Complications Following Total Hip Arthroplasty” https://www.intechopen.com/books/arthroplasty-update/complications-following-total-hip-arthroplasty
3) “Complications of total hip arthroplasty” https://www.uptodate.com/contents/complications-of-total-hip-arthroplasty
4) “THA Other Complications” https://www.orthobullets.com/recon/5030/tha-other-complications
5) “Causes of Painful THA” https://hipandkneebook.com/complications
6) “Periprosthetic joint infection: Current concept” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3601222/
7) “Joint Replacement Infection” https://orthoinfo.aaos.org/en/diseases–conditions/joint-replacement-infection/
8) “Preventing Blood Clots After Hip or Knee Replacement Surgery or Surgery for a Broken Hip” https://www.ncbi.nlm.nih.gov/books/NBK107165/#:~:text=DVT%20is%20the%20most%20common,or%20weeks%20after%20the%20surgery.
9) “PREVENTION AND TREATMENT OF BLOOD CLOTS AFTER HIP AND KNEE REPLACEMENT SURGERY” https://www.stoptheclot.org/about-clots/toolkit-for-knee-hip-replacement-patients/prevention-and-treatment-of-blood-clots-after-hip-and-knee-replacement-surgery/
10) “Deep Vein Thrombosis” http://www.ucihealth.org/medical-services/orthopaedics/hip-knee-surgery-services/deep-vein-thrombosis
11) “BLOOD CLOTS IN ORTHOPEDIC SURGERY FACT SHEET” https://www.stoptheclot.org/about-clots/toolkit-for-knee-hip-replacement-patients/orthopedic-surgery-fact-sheet/
12) “Blood Clots After Surgery” https://www.webmd.com/dvt/blood-clots-after-surgery
13) “Hip Dislocation” https://www.hss.edu/condition-list_hip-dislocation.asp
14) “Dislocation Following Total Hip Replacement” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4298240/
15) “Risk factors for dislocation after primary total hip replacement: a systematic review and meta-analysis of 125 studies involving approximately five million hip replacements” https://www.thelancet.com/journals/lanrhe/article/PIIS2665-9913(19)30045-1/fulltext
16) “Hip replacement” https://www.mayoclinic.org/tests-procedures/hip-replacement/about/pac-20385042
17) “Total Hip Replacement Fundamentals” https://my.clevelandclinic.org/health/treatments/17102-hip-replacement
18) “Signs & Symptoms” https://www.sahortho.com/joint/signs-symptoms#:~:text=Thigh%20or%20groin%20pain%20is,a%20sign%20of%20implant%20loosening.
19) “Hip Replacement Loosening Symptoms” https://www.verywellhealth.com/hip-replacement-loosening-2548623#:~:text=The%20most%20common%20cause%20of,This%20problem%20is%20called%20osteolysis.
20) “Revision Total Hip Replacement: An Overview” https://www.hss.edu/conditions_revision-total-hip-replacement-overview.asp
21) “Dislocation after THA remains a dreaded complication for patients and surgeons” https://www.healio.com/news/orthopedics/20160525/dislocation-after-tha-remains-a-dreaded-complication-for-patients-and-surgeons
22) “Fracture After Total Hip Replacement” http://www.ucihealth.org/medical-services/orthopaedics/hip-knee-surgery-services/fracture-total-hip-replacement
23) “Periprosthetic Fractures of the Femur After Hip Arthroplasty: An Analysis of 99 Patients” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2504263/
24) “Fractures around a hip replacement or knee replacement” https://uihc.org/health-topics/fractures-around-hip-replacement-or-knee-replacement
25) “Total hip arthroplasty.” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1022709/
26) “Management of the Infected Total Hip Arthroplasty” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5525520/
27) “Hospitalization for Total Hip Replacement Among Inpatients Aged 45 and Over: United States, 2000–2010” https://www.cdc.gov/nchs/products/databriefs/db186.htm
28) “Prevalence of Total Hip and Knee Replacement in the United States” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4551172/
29) “Metal-on-Metal Hip Implants” https://www.fda.gov/medical-devices/implants-and-prosthetics/metal-metal-hip-implants#:~:text=To%20date%2C%20there%20are%20no,for%20use%20in%20the%20US.&text=Some%20patients%20who%20had%20a,%2Don%2Dmetal%20hip%20implant.
30) “Metal-on-Metal Hip Implants: The FDA’s Activities” https://www.fda.gov/medical-devices/metal-metal-hip-implants/metal-metal-hip-implants-fdas-activities
31) “Effectiveness of Metal-on-Metal Hip Implants” https://www.fda.gov/medical-devices/metal-metal-hip-implants/effectiveness-metal-metal-hip-implants
32) “Information about Soft Tissue Imaging and Metal Ion Testing” https://www.fda.gov/medical-devices/metal-metal-hip-implants/information-about-soft-tissue-imaging-and-metal-ion-testing
33) “Revision Total Hip Replacement” https://orthoinfo.aaos.org/en/treatment/revision-total-hip-replacement/
34) “Hip Revision Surgery Fundamentals” https://my.clevelandclinic.org/health/treatments/17104-hip-revision
35) “Product Classification” https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfPCD/classification.cfm?ID=LPH
36) “Hip Revision” https://www.hss.edu/condition-list_hip-revision.asp
37) “About Revision Hip Surgery” https://www.cedars-sinai.org/health-library/diseases-and-conditions/r/revision-hip-surgery.html
38) “What patients need to know about revision surgery after hip or knee replacement” https://www.sciencedaily.com/releases/2013/12/131231132730.htm
39) “Total Hip Arthroplasty: Past, Present, and Future. What Has Been Achieved?” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6892902/
40) “A look ahead to the future of hip replacement and hip resurfacing” http://www.opnews.com/2019/01/future-of-hip-replacement-and-hip-resurfacing-article/15075
41) “COMPLICATIONS OF HIP & KNEE REPLACEMENT” http://newyorkorthopedics.com/ny-orthopedics-doctors-highlights/complications-of-hip-knee-replacement-surgery-and-revision-surgery-options/
42) “Joint Replacement Surgery & Infection Treatment” https://www.newyorkhipknee.com/conditions/joint-infection/
43) “Complications of Hip Replacement Surgery” https://www.verywellhealth.com/problems-with-hip-replacement-surgery-2549564